Half life of carbon 14 formula3/9/2024  
Suppose we have 100.0 g of tritium (a radioactive isotope of hydrogen). The half-life of a specific radioactive isotope is constant it is unaffected by conditions and is independent of the initial amount of that isotope.Ĭonsider the following example. 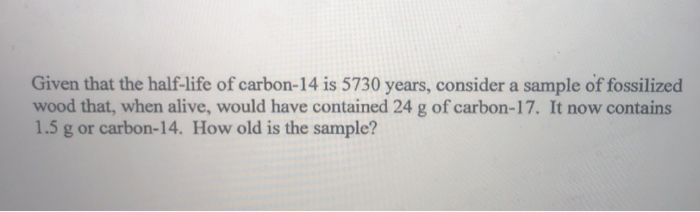
An interesting and useful aspect of radioactive decay is half-life, which is the amount of time it takes for one-half of a radioactive isotope to decay. As time passes, less and less of the radioactive isotope will be present, and the level of radioactivity decreases. Some isotopes are stable indefinitely, while others are radioactive and decay through a characteristic form of emission. Whether or not a given isotope is radioactive is a characteristic of that particular isotope. Determine the amount of radioactive substance remaining after a given number of half-lives. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
